Layperson terms for ketone body metabolism?
Layperson Terms | Auto-update medical terms | Low-carb community interest | Reactive hyperglycemia vs hypoglycemia | Bio-individuality & general terms | Metabolic illnesses defying general terms | Ketone Metabolism Disorders | Disentangling ketosis & ketoacidosis | Lead-thinker insights | Richard Feinman | Kristin Baier | Dave Feldman | Gabor Erdosi | Kristi Storoschuk | Josephine Barbarino | Siobhan Huggins | Viktoria Terman | John Madany | Sources | More by Mani Malagón | Other articles that may interest you
Can we disentangle ketosis & ketoacidosis?
The underlying difference between ketosis and ketoacidosis is healthy or faulty metabolism. (Pastromas 2008) In ketosis and ketonemia, fatty acids are converted to efficient ketone bodies to fuel brain, heart, kidney and muscle cells. On the other hand, in ketoacidosis damaged metabolic processes regulating blood glucose and ketone body levels have run awry. Ketosis and ketoacidosis represent opposite ends of the health spectrum.
Is the difference between ketosis, ketonemia & hyperketonemia only important to academics?
This first draft of layperson definitions hopes to promote a discussion eliciting biochemical and medical points-of-view from leading-thinkers in the low-carb and medical communities. The aim is improving layperson terms involving the benefits and potential hazards of metabolically disrupted ketone and glucose interactions.
Low blood glucose, hypoglycemia, can be asymptomatic?
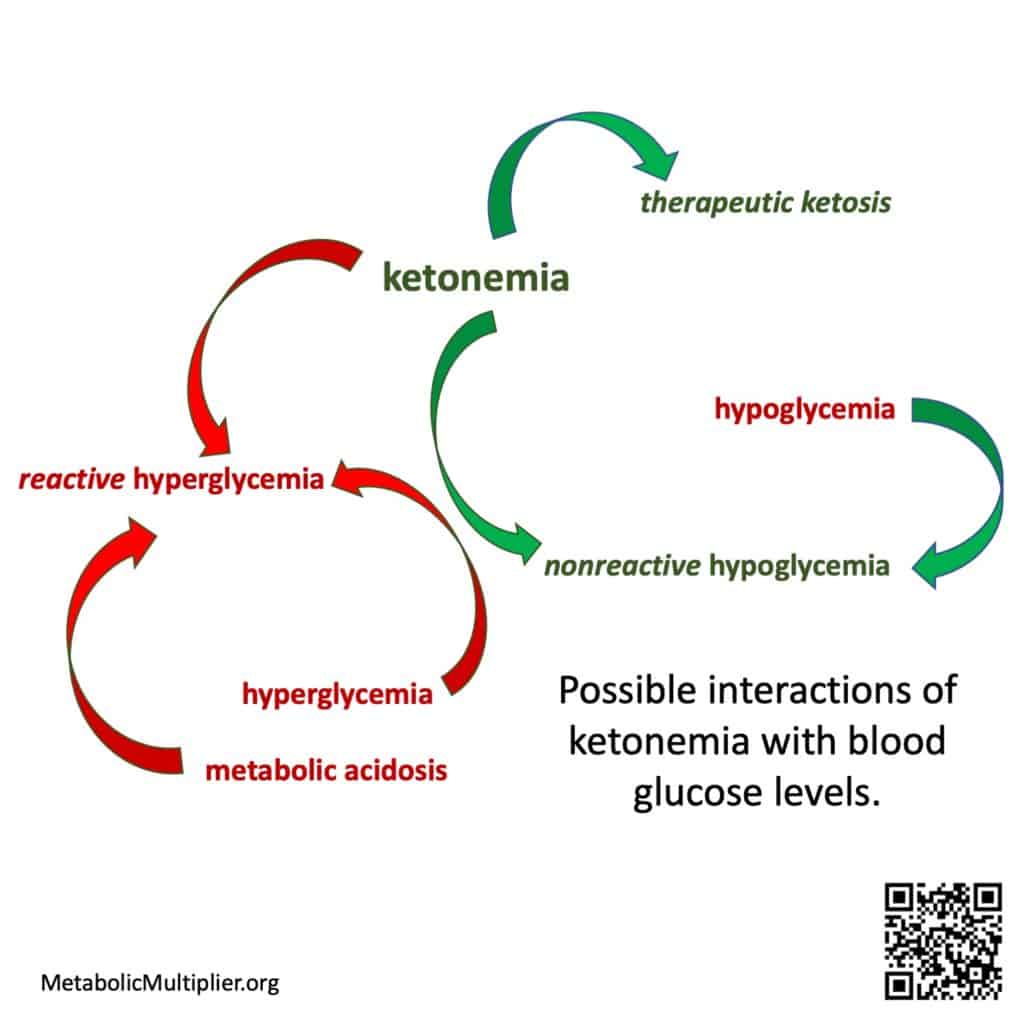
Wide blood glucose level swings due to metabolic illness such as diabetes, hyperglycemia to hypoglycemia, are a risk factor for cardiovascular disease. (Monnier 2012). Additionally, blood sugar levels dropping precipitously after consuming refined carbohydrate laden meals, postprandial hypoglycemia, can lead to acute and severe symptoms. However, when ketone bodies are present, the brain, heart and muscles can use these energy substrates instead of glucose resulting in asymptomatic non-reactive hypoglycemia. (Drenick 1972). Therefore, blood sugar levels and ketone body levels interact in beneficial ways in healthy individuals to maintain energy homeostasis.
So why is hypoglycemia so feared?
When metabolic dysfunction leads to unregulated generation of ketone bodies above physiological levels, hyperketonemia, coupled with hyperglycemia and acidosis, a dangerous ketoacidosis can result. (Kitabchi 2001). Diabetic ketoacidosis might be caused by poor insulin control in Type 1 Diabetes, but it can also be seen in Type 2 Diabetes and is often associated with infections (Casqueiro 2012)(Li 2020)(Rawla 2017). In these cases ketone bodies and glucose are improperly regulated due to underlying illness. That being said, if ketosis is conflated with ketoacidosis, then low-carbohydrate diets that often result in dietary ketosis might be considered dangerous.
Layperson terms for ketone metabolism
- ketone bodies (human) = acetone, acetoacetate (ACA), ß-hydroxybutyrate (ß-HB)
- ketogenesis = liver hepatocyte, gut microbiome, or brain astrocyte production of ACA or ß-HB from fatty acids. Liver production is usually in a ratio of 1-part ACA to 10-parts ß-HB. Ketogenesis is a means of transforming fatty acid energy substrates to ß-HB for distribution to other parts of the body where it is used as acetyl-coenzyme A (acetyl CoA). E.g., in the liver: (2) acetyl CoA —> ß-HB, which can then cross the blood-brain-barrier.
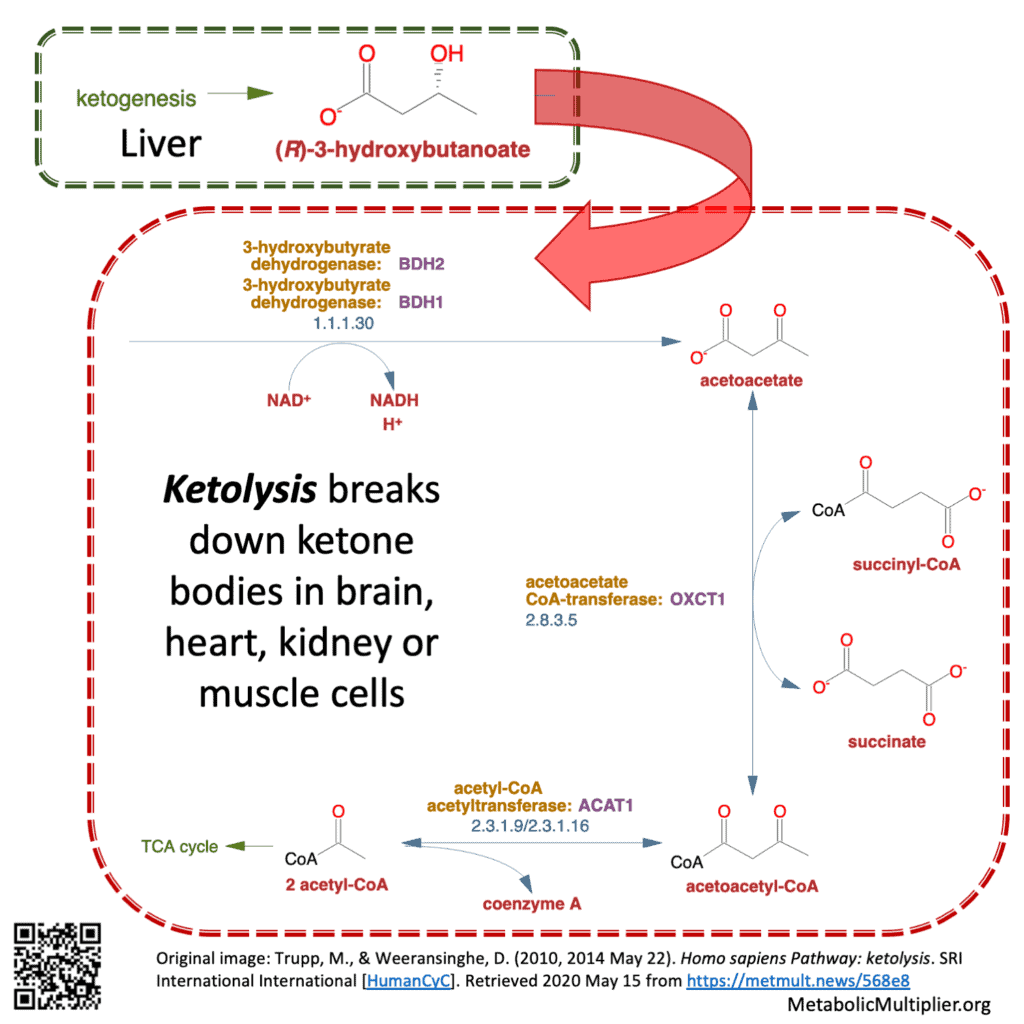
- ketolysis = breaking down of ketone bodies. E.g., in brain, muscle, kidney cells: ß-HB —> (2) acetyl CoA
- ketosis = measurable blood ketone bodies
- nutritional ketosis = measurable ketone bodies, usually > 0.2 mmoL/L
- ketonemia = elevation of nutritional blood ketone body levels to fasted-state blood ketone levels, e.g. ≥ 1.0 mmoL/L after 3-days fasting.
- Glucose/Ketone Index (GKI) = Blood glucose (mmol/L)/Blood ketones (mmoL/L) (Meidenbauer 2015)
- therapeutic ketosis = ketonemia without acidosis, usu. GKI ≤ 1.0 (Meidenbauer 2015)
- hyperketonemia = ketone body levels > 10 mmol/L (Kanikarla-Marie 2016)(added 23 May 2020)
- metabolic acidosis = blood pH < 7.3 (Gomez-Arbelaez 2017)(Noyes 2007). This is an acute condition.
- ketoacidosis = hyperketonemia + metabolic acidosis. This is an acute condition.
- hypoglycemia = blood glucose ≤ 40 mg/dL
- reactive hypoglycemia = hypoglycemia without ketosis. This is an acute condition a.k.a non-ketotic hypoglycemia. (Fukao 2014)
- nonreactive hypoglycemia = hypoglycemia + ketonemia.
- hyperglycemia = blood glucose ≥ 100 mg/dL
- reactive hyperglycemia = hyperglycemia + ketoacidosis. (e.g., in T1D as diabetic ketoacidosis with dehydration.) This is an acute condition that can lead to coma or death.
- nonreactive hyperglycemia = hyperglycemia without ketoacidosis. Chronic glucotoxicity leading to micro/macro-vascular damage (Chapin 2015), neurological damage, neuropathy, nephropathy, pancreatic ß-cell damage, Alzheimer’s, Parkinson’s, etc.
In fasting or starvation the liver makes ketone bodies from fatty-acids through ketosis (ketogenesis) an anabolic (building) process. Ketone bodies are the preferred fuel of the heart & brain. When glucose is low ketone bodies are readily broken down by cells through ketolysis, a catabolic (breaking-down) process, to provide energy substrates.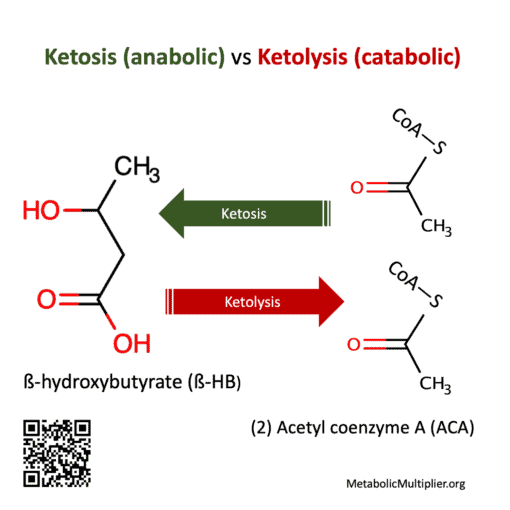
Only if ketosis is improperly thought to be a synonym for ketoacidosis or if they have a rare contraindication such as a fatty-acid or ketone metabolism disorder. If you are on a prescription medication or have a medical condition then you should include a competent medical health professional on your low-carbohydrate journey.
In type 1 diabetics or individuals with liver or kidney damage experiencing hyperglycemia, a high level of ketone bodies in the blood can result in metabolic acidosis and ketoacidosis.
Ketone bodies are the preferred fuel of the heart and brain. When glucose levels drop ketone bodies are readily used by other parts of the body and can prevent hypoglycemia symptoms. (Drenick 1972)
Where is the auto-update for medical terms?
We’ve been struggling with the word ketosis a century?
In 1917 Dr. Frederick M. Allen, MD stated that definitions require “need and distinctiveness. Diminished alkalinity, increased hydrogen ion concentration, lowering of carbon dioxide, decrease of buffer salts, and (for the symptoms of these changes) acid intoxication—all these terms have definite meanings, and to appropriate the name acidosis for any one of them is merely to create a useless synonym. No other name but acidosis exists for the metabolic process which it denotes. Ketonuria and ketonemia have their accurate place but do not cover the ground. Possibly the word ketosis might be suggested and used for special purposes, but the change of established usage would be difficult and seems unnecessary. (Allen 1917)[Added emphasis.]
Why no consistent ketone metabolism layperson definitions?
The English language is rapidly burgeoning with technical terms. (In contrast, the Spanish Royal Academy of Language tightly controls Spanish vocabulary). No one can keep up with all the terms. An unintended consequence of this rapid flood of vocabulary is the eddies, of still waters, on the margins. In those backwaters terms stagnate by not moving in consonance with the onrush. This may not be important for arcane terms, but in medical practice precision in terminology could mean life or death. An internet search of several current medical references [from the Merck Manual Professional to the NIH MeSH database] revealed that the words ketosis and ketoacidosis are either not described or are conflated.
Should the low-carb community care about layperson terms for ketosis?
Curating terminology based on precisely measured and quantified inputs (e.g., dextrose or sucrose?) and outputs (anthropometric, physiometric & biochemical biomarkers) might help settle the definition and effects of a low-carbohydrate diet (e.g., sugar or starch?) and a low-fat diet (e.g., Ω-3 or Ω-6?). Coherent definitions might change how we view occlusive terms like high-fat diet (HFD). For instance, is the current HFD definition of rodent chow [35% sugar & starch with 45% fat] adding value to diet wars that consider fat evil? How can high-sugar and high-fat (HSHF) rat chow lead to meaningful laboratory experiments if the food is a dietary mix? If ketosis is conflated with ketoacidosis, doesn’t that mean your doctor might panic when you tell her you’re in ketosis?
What is reactive hyperglycemia or reactive hypoglycemia?
This initial draft of a tree of interrelated terms is glucose- & ketone- centric, i.e., the definitions exclude insulin’s regulatory effects and don’t delve into the anion gap in metabolic acidosis. The attempt here is to provide a set of layperon definitions in the hope of prompting a wider discussion by the medical community to disentangle ketosis and ketoacidosis. The hope is to highlight the protective effects of ketonemia when blood sugar levels drop resulting in nonreactive hypoglycemia (Drenick 1972)(McCall 1982)(Rehni 2018); and, contrast that situation with the one where damaged kidney or liver function is aggravated with hyperglycemia resulting in ketoacidosis.
But what about bio-individuality and these general terms?
We dreamed of personalized measurements
Integration of human genome data with clinical, biochemical, physiological and other data obtained both directly from humans and indirectly from model organisms should, over time, lead to a deeper understanding of human metabolism and its nutritional implications in health and disease. When the genome sequences of individuals are available, it may be possible to address questions about the variation in optimal nutrition from person to person.
(Romero 2004)
We have blood glucose & ketone meters
The importance of portable devices in the treatment of hyperglycemia and hyperketonemia (diabetic ketoacidosis – DKA) was dramatically demonstrated in a study with T1D children. Two endpoints were set for measuring successful DKA treatment by an “integrated care pathway (ICP) with fluid replacement and insulin infusion”:
- DKA ICP A: pH > 7.3 with two successive ß-HB readings < 1 mmol/L
- DKA ICP B: pH > 7.3 and no ketonuria (urine ketone body free) [this was the current endpoint.]
The results indicated that both endpoints worked since in “35 patient episodes, the ICP was completed (28 to negative ketonuria) without significant variation.” However, the time to intervention was much earlier for DKA ICP A!
- DKA ICP A: reached after 17 h (4-39 h)
- DKA ICP B: not reached until 28 h (14-64 h) after starting treatment
These results lead to reduced insulin doses in the treatment of diabetic ketoacidosis!
(i) Serial measurement of NPT HOB [near-patient testing meter ß-HB] allows evaluation of a new, simple, earlier end-point for intravenous insulin therapy.
(Noyes 2007)
(ii) Agreement between NPT and laboratory HOB was clinically acceptable for HOB levels within the meter’s analytical range.
What are examples of metabolic illnesses that defy general terms?
Here are some examples of fatty acid metabolic diseases that show the complexity that the human body can manifest. Notice that in some of these cases individuals must avoid fasting because they have a malfunction that does not allow them to burn fat!
Ketone Metabolism Disorders
| Disease (OMIM Number) | Defective Proteins or Enzymes | Comments |
| 3-Hydroxy-3-methylglutaryl-CoA synthase-2 deficiency (605911*) | 3-Hydroxy-3-methylglutaryl-CoA synthase | Biochemical profile: See below Clinical features: Episodic nonketotic hypoglycemia, encephalopathy, hepatomegaly Treatment: Avoidance of fasting |
| 3-Hydroxy-3-methylglutaryl-CoA lyase deficiency (246450*) | 3-Hydroxy-3-methylglutaryl-CoA lyase | Biochemical profile: Hypoglycemia, metabolic acidosis without ketonuria Clinical features: Irritability, lethargy, vomiting Treatment: Avoidance of fasting, leucine restriction |
| Succinyl-CoA:3-oxoacid-CoA transferase deficiency (245050*) | Succinyl-CoA 3-oxoacid-CoA transferase | Biochemical profile: Ketonuria Clinical features: Severe episodic ketoacidosis, vomiting, hyperventilation Treatment: Glucose during acute episodes plus judicious use of bicarbonate, high-carbohydrate diet with some restriction of protein and fat |
| Mitochondrial acetoacetyl-CoA thiolase deficiency (203750*) | Mitochondrial acetoacetyl-CoA thiolase | Biochemical profile: Severe metabolic acidosis, hypoglycemia, ketonuria Clinical features: Lethargy, vomiting Treatment: Avoidance of fasting |
| Cytoplasmic acetoacetyl-CoA thiolase deficiency (614055*) | Cytoplasmic acetoacetyl-CoA thiolase | Biochemical profile: Ketonuria Clinical features: Intellectual disability, hypotonia Treatment: Not established |
| (Demczko 2020) |
Leading-thinker insights:
Some of the low-carb movement’s thought-leaders have been kind enough to share the following insights as they reviewed the facts underlying these layperson definitions:
Richard Feinman, Ph.D., Feinman the Other
“In specific biochemical, medical cases, you want to be careful about a priori cut-and-dried definitions. The context determines everything. Even ‘ketones.’ There are literally an infinite number of ketones and in our context it is okay to use the term to mean ketone bodies. In talking to organic chemists there are other keto-acids in biology (in degradation of branch chain amino acids) — it can be imprecise.
You are right to take on the confusion — surprisingly, more wide spread than I would have guessed, between ketosis and ketoacidosis. But you have to be careful how you explain it, that is, over-explain it.” (Richard D. Feinman, PhD, personal communications, 19 May 2020).
Kristin Baier, M.D. Calibrate
“Emphasizing the distinction of nutritional ketosis and diabetic ketoacidosis is imperative. Using the complete terms, for example adding nutritional in front of ketosis every time when discussing a ketogenic diet, or changing the dialogue to state non-acidotic nutritional ketosis instead of simply saying ‘ketosis.'”
Secondly, is ketosis the same as ketoacidosis? “No! The term ketosis does not imply acidosis is present. Distinguishing that non-acidotic nutritional ketosis is not the same as ketoacidosis is crucial. Sadly most physicians were only taught ketoacidosis and have no idea any other forms of ketosis exist.” — (Kristin Baier, MD, personal communication, 22 May 2020)[Added emphasis.]
Dave Feldman, Cholesterol Code:
“This concise summary distinguishes important differences in biomarkers between ketosis for metabolic homeostasis vs those of clear concern showing energy toxicity and dysregulation. It is long overdue. Thank you for helping to advance these key distinctions.” – (Dave Feldman [Cholesterol Code], personal communication, 22 May 2020.)
Gabor Erdosi, Meterbolic:
“I believe that this topic, though complex, does not merit constant confusion. Apart from a few rare genetic diseases and a handful of drug overdose (or side-) effects:
- Ketoacidosis results from uncontrolled ketosis, typically as the result of absolute insulin deficiency;
- The root physiological cause is uncontrolled lipolysis, i.e. low insulin and high free-fatty acid (FFA) flow to the liver;
- In physiological ketosis insulin is low, but it is still sufficient to limit FFA outflow and protein catabolism at appropriate levels;
- Low insulin stimulates both high ketogenesis (ketonemia) and high gluconeogenesis (hyperglycemia); which are further exacerbated by protein catabolism due to insulin deficiency;
- Consequently, without insulin, ketoacidosis results from non-physiological ketones (above ~8 mmol/L) coupled with non-physiological glucose (also above ~8 mmol/L);
- i.e., in the absence of insulin, the end result is an uncontrolled trajectory of ketone and glucose overproduction leading to ketoacidosis;
- Opposing trajectories of glucose and ketones is part of healthy physiology, not ketoacidosis. This can be nicely observed when a healthy Average Joe does a multi-day fast. Ketones gradually increase to ~7 mmol/L while glucose gradually drops to ~3 mmol/L.
- p.s. As a side note, “I’m about to launch lowerinsulin.com … Since the “brand” has been out there as the Lower Insulin Facebook group and page.” — (Gabor Erdosi, personal communication, 22 May 2020)[Added emphasis.]
Kristi Storoschuk, Science Journalist:
- “I would add for nutritional ketosis that it is diet-induced ketosis (ß-HB usually falls within 0.5-3.0 mmol/L), in contrast to starvation or fasting induced ketosis (ß-HB usually between 3.0 and 7.0 mmol/L), with ketoacidosis occurring when ketones reach >10.0 mmol/L in combination with high blood glucose. With even more nuance being added by exogenous-ketone induced ketosis and exercise-induced ketosis.
- The 4 major routes of achieving ketosis are: diet, fasting, exogenous ketones, and exercise.
- The main idea here is that “ketosis” is one definition, the strategies of achieving it have different definitions, and based on the strategies ß-HB may fall into different ranges.
- Some mention of a negative feedback mechanism via insulin would help people understand why healthy individuals (non-T1D) are protected from ketoacidosis. Since insulin regulates ketone production, pathologically high blood ketones (>10.0 mmol/l) are due to insulin insufficiency.” — (Kristi Storoschuk, personal communication, 22 May 2020)[Added emphasis.]
Josephine Barbarino, Keto Live Project
“I think it is very important to clarify and unify these questions! Not only for patients, to arm them with the knowledge to interact with professionals and to be able to make informed decisions; but equally for professionals, who often have their information heavily influenced by Big Food and Big Pharma.” (Josephine Barbarino, personal communication, 22 May 2020).
Siobhan Huggins, Cholesterol Code & Own Your Labs
“Regarding glucotoxicity above 100 mg/dL glucose, two examples come to mind of apparently, or possibly, benign non-reactive hyperglycemia. One in a ketogenic state, and the other not.
1. A non-low carb example of apparently generally benign non-reactive hyperglycemia is persons with heterozygous glucokinase mutations. They have diabetic levels of steady state hyperglycemia throughout their lifetime (running hba1c levels of 5.8-7.6), but do not generally present with hyperinsulinemia and micro/macrovascular complications are reportedly rare. (Chakera 2015) [https://care.diabetesjournals.org/content/38/7/1383].
2. Lean Mass Hyper-responders (LMHR) or those on very low carb ketogenic diets with HDL of >80 mg/dL, LDL of >200 mg/dL, and triglycerides of <70 mg/dL and usually have lean bodies. Although not all LMHRs have higher glucose levels, a subset (usually those are more frequently active/heavier exercisers) can sometimes present with higher fasting glucose levels and hbA1c (typically even milder than the glucokinase example) paired also with lower insulin (usually <5 uIU/mL). However, based on anecdotal reports, when LMHR glucose is monitored via a Continuous Glucose Monitor (CGM) the variation shows steady state glucose from food, but higher levels either in the mornings and especially while exercising. However, this isn’t confirmed to be benign, and further study on LMHRs with this presentation is needed to further elucidate if it presents with increased risk for hyperglycemia associated complications.
Thus, instead of pairing it without ketoacidosis I would instead pair it with hyperinsulinemia. This would help clarify the pathological context that would pair with the conditions you specified, the two above examples outline the without hyperinsulinemia scenario. Another difference is steady state glucose (no large up and down spikes from food) or absent metabolic syndrome [as also mentioned in the heterozygous glucokinase mutation paper (Chakera 2015)].” (Siobhan Huggins, personal communications, 27 May 2020)[Original emphasis, added color.]
Viktoria Terman, Instructor Surgical Technology
“The anion gap is measured by subtracting anions from cations. Normal anion gap levels are ~3 to 5 mEq/L. Potassium (K+) is affected by insulin. Hyperglycemia leads to hypokalemia. This is because as insulin levels rise, K+ levels in the intra-cellular fluid decrease as K+ moves out of the cell, thereby increasing the anion gap and lowering potassium levels in cells.
T1D’s lack insulin and can have both hyperglycemia and hyperketonemia, which results in diabetic ketoacidosis (DKA). In patients with DKA the anion gap changes due to hyperglycemia with no insulin available. Or, in the case of insulin resistance (T2D) it changes due to compensatory insulin secretion with no effect on hyperglycemic levels. Either situation decreases HCO3– (bicarbonate) and furthers acidosis. Ketoacidosis is a pathological metabolic condition.
Lastly, one of the worst ravages of hyperinsulinemia and hyperglycemia are their damage to fibrinolysis and activation of coagulation factors leading to endothelial damage.” (Viktoria Terman, personal communication, 22 May – 1 June 2020). [Added emphasis. mEq/L = milliequivalents/liter, anion gap = (Na+ + K+) – (Cl– + HCO3–), hypokalemia = low potassium (K+) levels](Stegenga 2006).
Dr John Madany, MD Barret Hospital & Health Care
“Medical education does not distinguish ketosis from ketoacidosis in any significant way. In medical education ketosis may be mentioned as the body having an alternate fuel supply but a little more. Ketoacidosis is something that requires emergency medical care and has a significant mortality rate. From a medical training perspective doctors definitely associate ketones with a negative connotation.”
Secondly, is ketosis the same as ketoacidosis? “Definity not, but as a consequence of modern dietary habits ketosis has become “abnormal “. Ketosis is frequently seen in conjunction with acute illness, correlating with illness. Ketosis may be found in a diabetic early in the progression to DKA. Now that ketones can be monitored at home, I have seen pre-DKA and successfully treated it in the outpatient setting.
A recent study aptly states that a “well-formulated” ketogenic diet is generally characterized by a total carbohydrate intake of less than 50 g/d and a moderate protein intake of approximately 1.5 g/d per kg of reference weight. This typically increases circulating ß-hydroxybutyrate (BHB) and acetoacetate (ACA) from concentrations that are typically less than 0.3 mM into the range of nutritional ketosis, which for BHB, we define as 0.5–3 mM). This range is below the typical 5–10 mM range for BHB that occurs during prolonged fasting, and well below concentrations characteristic of ketoacidosis. (Volek 2011)(Miller 2018).” (Dr John Madany, MD, personal communication, 1 June 2020.)[Additional emphasis.]
Sources on ketone metabolism:
Allen, F. M. (1917). The role of fat in diabetes. In G. M. Piersol & J. H. Musser (Eds.), The American Journal of the Medical Sciences (Vol. CLIII, pp. 313-371). Lea & Febiger. https://metmult.news/46427
Casqueiro, J., Casqueiro, J., & Alves, C. (2012). Infections in patients with diabetes mellitus: A review of pathogenesis. Indian journal of endocrinology and metabolism, 16 Suppl 1(Suppl1), S27-S36. https://doi.org/10.4103/2230-8210.94253
Chakera, A. J., Steele, A. M., Gloyn, A. L., Shepherd, M. H., Shields, B., Ellard, S., & Hattersley, A. T. (2015). Recognition and Management of Individuals With Hyperglycemia Because of a Heterozygous Glucokinase Mutation. Diabetes Care, 38(7), 1383-1392. https://doi.org/10.2337/dc14-2769
Crawford, P. A., Crowley, J. R., Sambandam, N., Muegge, B. D., Costello, E. K., Hamady, M., Knight, R., & Gordon, J. I. (2009). Regulation of myocardial ketone body metabolism by the gut microbiota during nutrient deprivation. Proceedings of the National Academy of Sciences of the United States of America, 106(27), 11276-11281. https://doi.org/10.1073/pnas.0902366106
Demczko, M. (2020 April) Ketone Metabolism Disorders [table] in Overview of Fatty Acid and Glycerol Metabolism Disorders, Merck Manual Professional Version, accessed 2020 May 20 at https://www.merckmanuals.com/professional/pediatrics/inherited-disorders-of-metabolism/overview-of-fatty-acid-and-glycerol-metabolism-disorders
Drenick, E. J., Alvarez, L. C., Tamasi, G. C., & Brickman, A. S. (1972). Resistance to symptomatic insulin reactions after fasting. The Journal of Clinical Investigation, 51(10), 2757-2762. https://doi.org/10.1172/JCI107095
Fukao, T., Mitchell, G., Sass, J. O., Hori, T., Orii, K., & Aoyama, Y. (2014). Ketone body metabolism and its defects. J Inherit Metab Dis, 37(4), 541-51. https://doi.org/10.1007/s10545-014-9704-9
Gomez-Arbelaez, et al. (2017). Acid-base safety during the course of a very low-calorie-ketogenic diet. Endocrine, 58(1), 81-90. https://doi.org/10.1007/s12020-017-1405-3 A very-low calorie ketogenic diet (VLCKD) does not cause acidosis.
Hassan, M. (2020). Acidosis and anion gap. In A. Takada (Ed.), New insights into metabolic syndrome. IntechOpen. https://doi.org/10.5772/intechopen.91760
Hess-Fischl, A. (2018 September 18). Hyperglycemia: When Your Blood Glucose Level Goes Too High, endocrineweb, accessed 17 May 2020, https://www.endocrineweb.com/conditions/hyperglycemia/hyperglycemia-when-your-blood-glucose-level-goes-too-high Hyperglycemia – fasting blood glucose > 130 mg/dL; postprandial (reactive) blood glucose >180 mg/dL.
Kanikarla-Marie, P., & Jain, S. K. (2016). Hyperketonemia and ketosis increase the risk of complications in type 1 diabetes. Free radical biology & medicine, 95, 268-277. https://doi.org/10.1016/j.freeradbiomed.2016.03.020
Kitabchi, A. E. (2001). Management of hyperglycemic crises in patients with diabetes. Diabetes Care, 24. https://doi.org/10.2337/diacare.24.1.131
Le Foll, C., & Levin, B. E. (2016). Fatty acid-induced astrocyte ketone production and the control of food intake. American journal of physiology. Regulatory, integrative and comparative physiology, 310(11), R1186-R1192. https://doi.org/10.1152/ajpregu.00113.2016
Li, J., Wang, X., Chen, J., Zuo, X., Zhang, H., & Deng, A. (2020). COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes Metab. https://doi.org/10.1111/dom.14057
McCall, A. L., Millington, W. R., & Wurtman, R. J. (1982). Metabolic fuel and amino acid transport into the brain in experimental diabetes mellitus. Proc Natl Acad Sci U S A, 79(17), 5406-5410. https://doi.org/10.1073/pnas.79.17.5406
Meidenbauer, J. J., Mukherjee, P., & Seyfried, T. N. (2015). The glucose ketone index calculator: a simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutrition & Metabolism, 12(1), 12. https://doi.org/10.1186/s12986-015-0009-2
Miller, V. J., Villamena, F. A., & Volek, J. S. (2018). Nutritional Ketosis and Mitohormesis: Potential Implications for Mitochondrial Function and Human Health. Journal of nutrition and metabolism, 2018, 5157645-5157645. https://doi.org/10.1155/2018/5157645
Monnier, L., Colette, C., & Owens, D. (2012). The glycemic triumvirate and diabetic complications: is the whole greater than the sum of its component parts? Diabetes Res Clin Pract, 95(3), 303-311. https://doi.org/10.1016/j.diabres.2011.10.014
Noyes, K. J., Crofton, P., Bath, L. E., Holmes, A., Stark, L., Oxley, C. D., & Kelnar, C. J. (2007). Hydroxybutyrate near-patient testing to evaluate a new end-point for intravenous insulin therapy in the treatment of diabetic ketoacidosis in children. Pediatr Diabetes, 8(3), 150-156. https://doi.org/10.1111/j.1399-5448.2007.00240.x
Pastromas, S., Terzi, A. B., Tousoulis, D., & Koulouris, S. (2008). Postprandial lipemia: an under-recognized atherogenic factor in patients with diabetes mellitus. Int J Cardiol, 126(1), 3-12. https://doi.org/10.1016/j.ijcard.2007.04.172
Rawla, P., Vellipuram, A. R., Bandaru, S. S., & Pradeep Raj, J. (2017). Euglycemic diabetic ketoacidosis: a diagnostic and therapeutic dilemma. Endocrinology, diabetes & metabolism case reports, 2017, 17-0081. https://doi.org/10.1530/EDM-17-0081
Rehni, A. K., & Dave, K. R. (2018). Impact of Hypoglycemia on Brain Metabolism During Diabetes. Molecular Neurobiology, 55(12), 9075-9088. https://doi.org/10.1007/s12035-018-1044-6
Romero, P., Wagg, J., Green, M. L., Kaiser, D., Krummenacker, M., & Karp, P. D. (2004). Computational prediction of human metabolic pathways from the complete human genome. Genome Biology, 6(1), R2. https://doi.org/10.1186/gb-2004-6-1-r2
Stegenga, M. E., van der Crabben, S. N., Levi, M., de Vos, A. F., Tanck, M. W., Sauerwein, H. P., & van der Poll, T. (2006). Hyperglycemia stimulates coagulation, whereas hyperinsulinemia impairs fibrinolysis in healthy humans. Diabetes, 55(6), 1807-1812. https://doi.org/10.2337/db05-1543
Trupp, M. (2010 April 20). Homo sapiens Pathway: ketogenesis. SRI International [HumanCyC]. Retrieved 2020 May 15 from https://metmult.news/91d4b
Trupp, M., & Weeransinghe, D. (2010, 2014 May 22). Homo sapiens Pathway: ketolysis. SRI International International [HumanCyC]. Retrieved 2020 May 15 from https://metmult.news/568e8
Umpierrez, G., & Korytkowski, M. (2016). Diabetic emergencies — ketoacidosis, hyperglycaemic hyperosmolar state and hypoglycaemia. Nature Reviews Endocrinology, 12(4), 222-32. https://doi.org/10.1038/nrendo.2016.15
Volek, J., & Phinney, S. D. (2011). The art and science of low carbohydrate living: An expert guide to making the life-saving benefits of carbohydrate restriction sustainable and enjoyable. Beyond Obesity.
More by Mani Malagón
- Diabetes Dirge, aka Diabetes for Dummies, learning to use big words to create positive health
- Ketosis versus ketoacidosis, metabolic flexibility or pancreatic disability?
- Pain started my ketogenic journey (Part 1)
- Coronavirus quarantine – how a lifetime in submarines eases my use of isolation for self-improvement
Other resource guides & toolkits you might value
Curious about improving metabolic health with lifestyle?
- Lifestyle Medicine, the key to metabolic healing
- What is metabolic health? A layperson’s definition and a biochemist’s perspective
Where do I start improving my metabolic health?
- Your patient toolkit for prediabetes & type 2 diabetes
- What to eat? Get clever! Clinicians’ flexible approach to tailoring carb reduction to you.
- Want more detail on carbohydrate reduction? A round-up of quality, what-to-eat guides from around the web
- Best resources for an overview of keto – Metabolic Multiplier’s round up of the best resources we’ve found by evidence-based organizations & clinicians. For patients, families & caregivers.
- Keto doctors near me – over 20 directories of low-carb professionals
For clinicians using therapeutic carbohydrate reduction (TCR), ketogenic diet therapies (KDT)
- Best Keto resources for clinicians & other practitioners
- How to build my ketogenic therapy treatment team – Support for the more complex cases.
- Why write medical case reports about unique cases? – Our round up can save you time.
For colleagues interested in amplifying metabolic health
- Overview schematics help us think about metabolic health – Understand our Organizing Research team‘s thinking behind these 3 metabolic health research portals
- Communication guide to unite advocacy for metabolic health – Suggested language metabolic health advocacy. Principles. Language to use or avoid. Food & lifestyle first.
- Style Guide to unite advocacy for metabolic health – Welcome new team members! Many of you encourage using one voice for metabolic health. Below are suggestions from your fellow clinicians, scientists and professionals. See our Communication Guide as well. We’d love to hear…
- What Foundations support Metabolic Health? Charities, advocacy & grassroots groups using metabolic interventions & health to treat metabolic conditions.











4 Responses
[…] Layperson terms for ketone body metabolism? […]
[…] Ketone body metabolism terms […]
[…] Metabolic Multiplier – 4.0 Metabolism || Layperson terms for ketone body metabolism || Ketosis versus ketoacidosis || Mani’s metabolic […]
[…] Ketone body metabolism terms […]